Amino acids
Amino acids are biologically important organic compounds made from amine and carboxylic
acid functional groups, along with a side-chain specific to each amino acid.
The key elements of an amino acid are carbon, hydrogen, oxygen, and nitrogen, although
other elements such as sulfur are found in the side-chains of certain amino acids.
In the form of proteins, amino acids comprise the second largest component - apart
from water - in human muscles, cells and other tissues.
Because of their biological significance, amino acids are essential in nutrition
and are commonly used in nutritional supplements.
Standard Amino Acids
Twenty amino acids are known as "standard" amino acids, which are encoded directly
from the genetic code.
Outside proteins, amino acids also perform critical biological roles including neurotransmitting,
transport, and synthesis of special compounds.
For example the standard glutamic acid and the non-standard gamma-amino acid: gamma-amino-butyric
acid (GABA) are the brain's main excitatory and inhibitory neurotransmitters, respectively.
Hydroxyproline - a major component of the connective tissue collagen - is synthesised
from proline.
The standard amino acid glycine is used to synthesise porphyrins used in red blood
cells, and the non-standard carnitine is used in lipid transport.
Essential Amino Acids
Nine of the 20 standard amino acids are called "essential" amino acids for humans
because they cannot be created from other compounds by the human body. These amino
acids, which must be present in the food, are: histidine, isoleucine, leucine, lysine,
methionine, phenylalanine, threonine, tryptophan and valine.
The remaining 11 non-essential amino acids, which can be made in the body from other
molecules, are: alanine, arginine, asparagine, aspartic acid, cysteine, glutamic
acid, glutamine, glycine, proline, serine and tyrosine.
Amino Acid Sructure
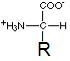 In the structure shown here, R represents a side-chain specific to each amino acid.
Depending on their side-chain the amino acids have different characteristics. The
side-chain can make an amino acid a weak acid or a weak base, and hydrophilic if
the side-chain is polar or hydrophobic if it is nonpolar.
In the structure shown here, R represents a side-chain specific to each amino acid.
Depending on their side-chain the amino acids have different characteristics. The
side-chain can make an amino acid a weak acid or a weak base, and hydrophilic if
the side-chain is polar or hydrophobic if it is nonpolar.
The hydrophobic amino acids tend to repel the aqueous environment and, therefore,
reside predominantly in the interior of proteins. These amino acids neither ionize
nor participate in the formation of H-bonds.
The hydrophilic amino acids tend to interact with the aqueous environment, they are
often involved in the formation of H-bonds and are predominantly found on the exterior
surfaces proteins or in the reactive centers of enzymes.
The amino acids with nonpolar hydrophobe aliphatic R-groups comprise glycine, alanine,
valine, 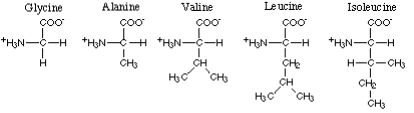 leucine and isoleucine.
leucine and isoleucine.
Of these valine, leucine and isoleucine have a branched chain R-group - they are
therefore termed "branched-chain amino acids" or BCAA. Here are the foods with the
highest content of glycine, alanine, valine, leucine and isoleucine.
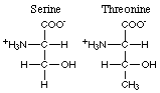 The non-aromatic amino acids with polar hydroxyl R-groups comprise serine and threonine.
Here are the foods with the highest content of serine and threonine.
The non-aromatic amino acids with polar hydroxyl R-groups comprise serine and threonine.
Here are the foods with the highest content of serine and threonine.
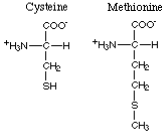 The amino acids with nonpolar sulfur-containing R-groups comprise cysteine and methionine.
Here are the foods with the highest content of cysteine and methionine.
The amino acids with nonpolar sulfur-containing R-groups comprise cysteine and methionine.
Here are the foods with the highest content of cysteine and methionine.
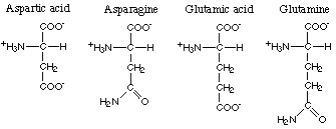 The amino acids with polar acid or corresponding polar amide R-groups comprise aspartic
acid, asparagine, glutamic acid and glutamine. Here are the foods with the highest
content of aspartic acid, asparagine, glutamic acid and glutamine.
The amino acids with polar acid or corresponding polar amide R-groups comprise aspartic
acid, asparagine, glutamic acid and glutamine. Here are the foods with the highest
content of aspartic acid, asparagine, glutamic acid and glutamine.
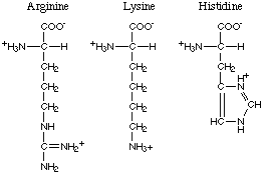 The amino acids with polar basic R-groups comprise arginine, lysine and histidine.
Here are the foods with the highest content of arginine, lysine and histidine.
The amino acids with polar basic R-groups comprise arginine, lysine and histidine.
Here are the foods with the highest content of arginine, lysine and histidine.
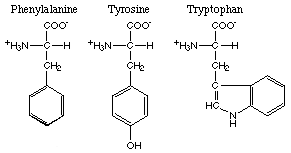 The amino acids with aromatic R-groups comprise phenylalanine, tyrosine and tryptophan.
Of these phenylalanine and tryptophan are nonpolar and tyrosine is polar. Here are
the foods with the highest content of phenylalanine, tryptophan and tyrosine.
The amino acids with aromatic R-groups comprise phenylalanine, tyrosine and tryptophan.
Of these phenylalanine and tryptophan are nonpolar and tyrosine is polar. Here are
the foods with the highest content of phenylalanine, tryptophan and tyrosine.
The amino acid with a nonpolar imino type R-group is proline. 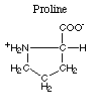 Here are the foods
with the highest content of proline.
Here are the foods
with the highest content of proline.
Amino Acid Function
It is the nature of the amino acid R-groups that dictate structure-function relationships
of peptides and proteins.
The hydrophobic amino acids will generally be encountered in the interior of proteins
shielded from direct contact with water.
Conversely, the hydrophilic amino acids are generally found on the exterior of proteins
as well as in the active centers of enzymatically active proteins. Indeed, it is
the very nature of certain amino acid R-groups that allow enzyme reactions to occur.
The imidazole ring of histidine allows it to act as either a proton donor or acceptor
at physiological pH. Hence, it is frequently found in the reactive center of enzymes.
Equally important is the ability of histidines in hemoglobin to buffer the H+ ions
from carbonic acid ionization in red blood cells. It is this property of hemoglobin
that allows it to exchange oxygen (O2) and carbon dioxide (CO2) at the tissues or
lungs, respectively.
The primary alcohol of serine and threonine as well as the thiol (–SH) of cysteine
allow these amino acids to act in enzymatic catalysis.
Additionally, the thiol of cysteine is able to form a disulfide bond with other cysteines.
This simple disulfide is identified as cystine.
The formation of disulfide bonds between cysteines present within proteins is important
to the formation of active structural domains in a large number of proteins.
Disulfide bonding between cysteines in different polypeptide chains plays a crucial
role in ordering the structure of complex proteins, e.g. the insulin receptor.
Rererences: 1 , 2
Copyright: Know Facts - All Rights Reserved




